We’ve arrived at a key early milestone in our research into Jordan’s Syndrome, a recently-discovered genetic mutation in the gene PPP2R5D thought to be linked to autism, Alzheimer’s, intellectual disability and cancer.
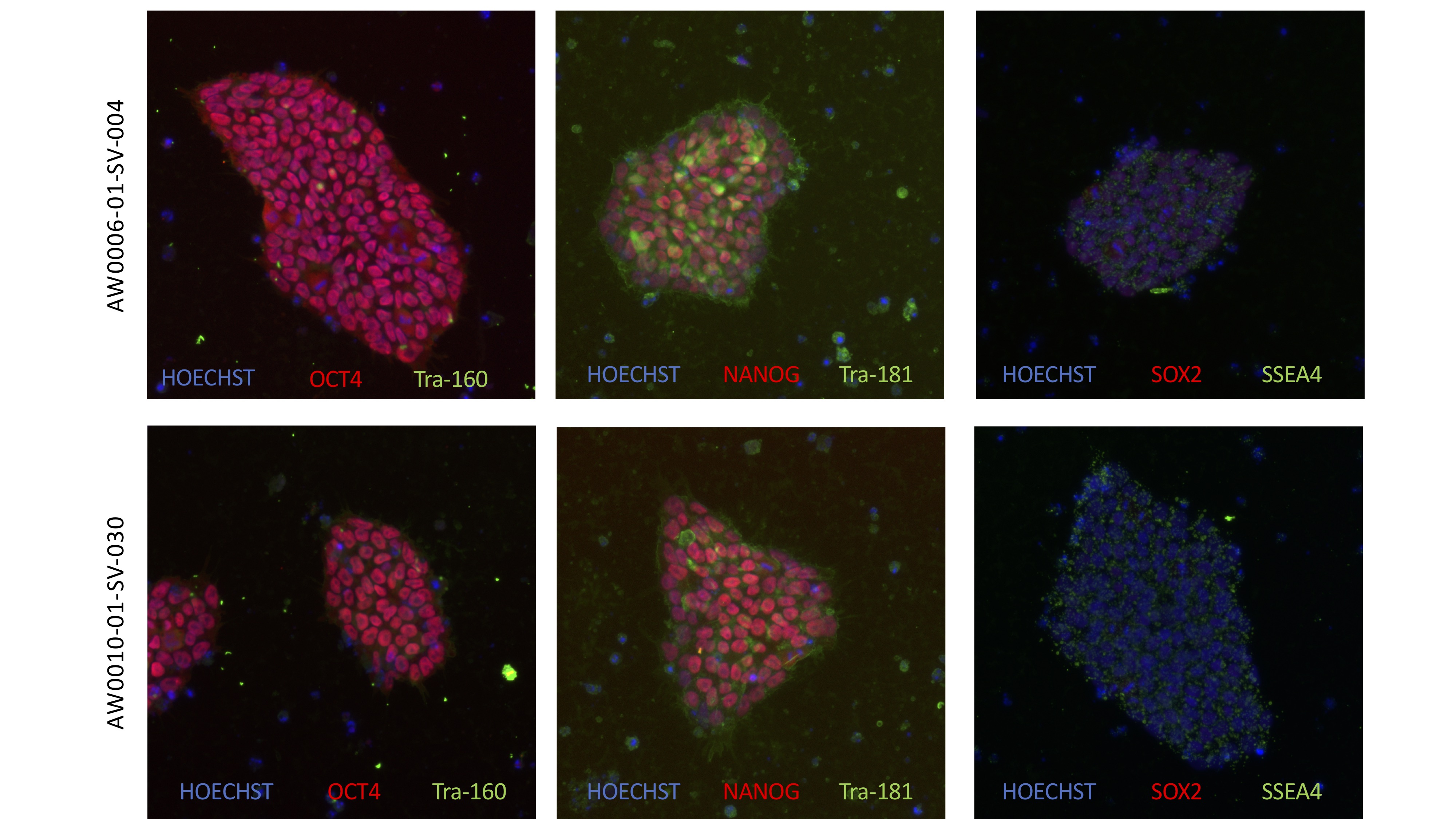
The first batch of iPSCs (induced pluripotent stem cells) have been created by the New York Stem Cell Foundation (NYSCF), and distributed to Rutgers University for duplication and distribution. The iPSCs were created from blood samples given by our children at the first family conference in Washington, D.C., and will play a critical role in understanding Jordan’s Syndrome – and how it may be reversed, treated or cured.
But – how exactly will that happen? And why are iPSCs so important? We asked our friends at the NYSCF to help explain.
How were the iPSCs created?
NYSCF: Scientists create induced pluripotent stem cells, or iPSCs, by taking a small amount of blood or skin from a patient, and converting these blood or skin cells into a pluripotent state using genetic signals called reprogramming factors. These pluripotent cells are able to multiply indefinitely and also can be coaxed into turning into any type of cell in the body, making them a unique and powerful research tool.
iPSCs enable scientists to study the cells affected by disease in the lab without harming patients. For example, the iPSC lines we are making in collaboration with Jordan's Guardian Angels will allow researchers to study patient-derived brain cells without brain surgery.
Because Jordan’s Syndrome is a rare disease with only a few dozen known patients worldwide, the stem cell lines we are making will provide a foundational resource for researchers who may not have easy access to patients. These patient-specific iPSC lines will be an invaluable tool for Jordan's Syndrome researchers around the world.
How many were created?
NYSCF: So far, NYSCF scientists have created five iPSC lines using blood samples from five different Jordan’s Syndrome patients. Each of these patients has a unique mutation in the gene implicated in the disease, PPP2R5D. Since the disease is so rare, these 5 lines represent a diverse segment of patients.
In addition to creating iPSC lines from additional patients, we are also planning to use stem cells to study the genetics of Jordan's syndrome. Using cutting-edge gene editing technology, our scientists will introduce PPP2R5D mutations into stem cells from a healthy individual. This 'manufactured disease line' will allow scientists to study the specific effects of disease-causing mutations, without being confounded by other differences between patients and healthy people that may be unrelated to the disease.
How can stem cells be used to find potential breakthrough treatments for Jordan’s Syndrome, and potentially related medical mysteries?
NYSCF: Stem cell technology allows scientists to create the different cell types affected in Jordan's Syndrome as patient-specific models of the disease. Modeling Jordan’s Syndrome in a dish using stem cell technology enables scientists to study how the disease develops in different cell types and affects their behavior over time. For example, scientists can derive patient neurons from stem cells, and even combine different types of patient brain cells into 'mini-brains' known as organoids, to study how brain function and the interactions between different cells are affected in Jordan's Syndrome. By enabling a better understanding of the disease and clarifying the cellular and molecular defects driven by the genetic PPP2R5D mutations, stem cell technology may point to treatments that can fix these defects and therefore alleviate disease symptoms.
Stem cell technology also allows new drugs to be directly tested on affected cells or organoids, to see if they have a beneficial effect. This "clinical trial in a dish" approach can help to identify new treatments that may be helpful for certain patients and ineffective – or even potentially toxic – to others.
The cellular processes that go awry in Jordan's Syndrome may well be playing a role in several other diseases – for example, the gene implicated in Jordan’s syndrome is also associated with autism, Alzheimer’s disease, and cancer. Stem cell technology has already advanced research into neurological diseases and cancer, just as we expect it will in Jordan's syndrome. A better understanding of what goes wrong in Jordan's Syndrome will help us understand how the processes in human development are supposed to work, and offer insights into other ways they go wrong in related diseases. There are mounting examples of rare disease research that have taught us something new about common diseases and how to treat them. So a new way to study the dozens of patients with Jordan's Syndrome could end up advancing knowledge and treatments for diseases that affect millions.
Give some perspective on how revolutionary this kind of science is, and its promise for medicine.
NYSCF: Stem cell research is a very young field. Embryonic stem cells were only first isolated in 1998, and the field drastically changed in 2006 with the development of induced pluripotent stem cells by Dr. Shinya Yamanaka, who received the Nobel Prize for this work just six years later. iPSCs made it possible to derive patient-specific cells, fueling the "disease-in-a-dish" and "clinical trial in a dish" research that has never been possible.
So the field has only really been in existence for just over a decade. It is truly the cutting edge of scientific inquiry with new discoveries, tools, and technology published every day. It is such an exciting time to be a stem cell researcher – as well as a disease researcher, because of the opportunities afforded by stem cell technology.
The promise of stem cell research is vast. At some point in our lives, we or the people we love become patients. Traditionally, much disease research and drug discovery have been performed using mouse models. But we are not mice, and to treat diseases more effectively we need to understand how diseases and drugs affect human cells – especially the types of cells involved in the disease. Taking brain cells out of patients to study them in the lab is not usually an option, but stem cell technology makes this type of research possible. The opportunity to create patient-specific stem cell lines has been revolutionary, because each patient is different, in what causes their disease, the way the disease manifests, and how they will respond to drugs. Stem cell research will therefore allow us to understand and treat disease in a more precise and personalized way than ever before possible.
Ultimately, our mission at NYSCF – and the goal in all of our collaborative stem cell projects – is to accelerate research towards cures for all devastating diseases, including Jordan’s Syndrome.